Failed Silicone ‘O’ Ring
Analytical Strategy:
This case study demonstrates how FTIR Analysis of a failed silicone ‘O’ ring can provide chemical compositional information that can determine the likely reason for its degradation.
Result
Comparison of the FTIR spectra of the failed ‘O’ ring (white powder) with that of an unused silicone ‘O’ ring (black rubber) shows that degradation of the organic silicone polymer results in destruction of the organic carbon part of the polymer. This results in an inorganic silica polymer residue that has no elasticity which is an essential property required for a rubber ‘O’ ring.
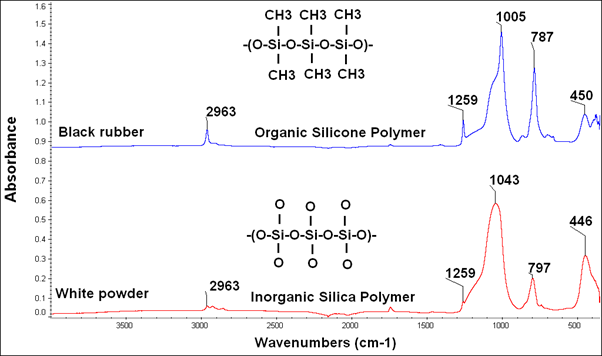
FTIR spectra
Conclusion
Based on the FTIR Analysis of the Failed Silicone ‘O’ Ring, the loss of a coherent carbon structure is probably due to oxidation caused by exposure to high temperature or a chemical reaction.

